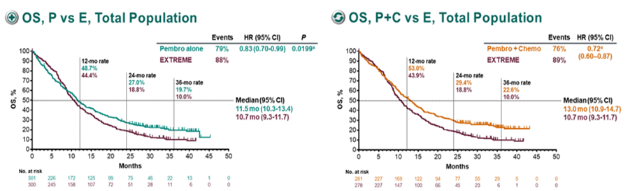
Keytruda also overtakes Opdivo in HNSCC, which at the moment is only approved as a second-line therapy for this form of cancer after chemotherapy in both the US and Europe. In Europe alone, it estimates there were more than 146,000 new cases and some 66,000 deaths from the disease last year. The combination regimen outperformed EXTREME regardless of PD-L1 expression levels.Īfter the KEYNOTE-48 results were published in The Lancet earlier this year, Keytruda alone and with chemo was billed as a “new standard of care” for HNSCC that represents “substantial progress for patients.”īMS says there were more than 705,000 new cases of head and neck cancer diagnosed worldwide in 2018, causing more than 358,000 deaths, so the new indication represents a big new market for Keytruda, already riding high with sales rising 62% to $3.1bn in the third quarter. In the trial, Keytruda as a monotherapy and in combination with chemo improved OS by 22% and 23%, respectively, compared to what has been the standard first-line treatment for this cancer for a decade – the EXTREME regimen of Eli Lilly’s Erbitux (cetuximab) with a platinum drug plus 5-FU – in the PD-L1-expressing group.Īs a monotherapy Keytruda proved to be non-inferior to EXTREME across all levels of PD-L1 expression, with a better safety profile and a response rate that was lower but much more long-lasting. Keytruda is also the first PD-1 therapy to improve overall survival in first-line HNSCC, hitting that objective in the phase 3 KEYNOTE-048 study reported last year. 
Nevertheless, Keytruda is the first anti-PD-1 drug to be cleared in Europe for this use, giving HNSCC patients a much-needed new treatment option and MSD’s drug another leg up over its rivals in the checkpoint inhibitor category such as Bristol-Myers Squibb’s Opdivo (nivolumab). The FDA approval was a little different, covering monotherapy with Keytruda (pembrolizumab) in PD-L1 expressors, but allowing the combination regimen to be used in ‘all-comers’, i.e. There are some cost-effectiveness studies related to these. The top-selling checkpoint inhibitor has been given the green light by the EMA for previously-untreated metastatic or unresectable recurrent head and neck squamous cell carcinoma (HNSCC) in patients whose tumours express PD-L1, both as a monotherapy and in combination with platinum and 5-fluorouracil (5-FU) chemotherapy. The inspiring results of KEYNOTE-048 might lead to pembrolizumab monotherapy and pembrolizumab in combination with chemotherapy becoming the first-line treatments for recurrent or metastatic HNSCC. The EMA has approved Merck & Co/MSD’s Keytruda as a first-line treatment for head and neck cancer, a few months after it was cleared by the FDA with a slightly broader label. Danny Rischin, MD, director, Division of Cancer Medicine, head, Department of Medical Oncology, Peter MacCallum Cancer Centre, discusses results from the protocol-specified final analysis of the phase III KEYNOTE-048 trial of pembrolizumab (Keytruda) as frontline therapy for recurrent/metastatic head and neck squamous cell carcinoma.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
