|
Materials appropriate for this application include boron carbide and the heavy metal cadmium. Schematic representation of a controlled nuclear chain reaction – note that the neutron absorbers block two of the fission pathways shown in the earlier figure.Ĭontrol rods must be fabricated from a material that readily absorbs or “captures” neutrons, but is not prone to nuclear fission as a consequence. a chain reaction is occurring), the control rods are inserted slightly further into the core in order to slow the rate of fission to the required value and maintain it at that level. To initiate a chain reaction when a reactor is being started up, the control rods are partially withdrawn so that they absorb fewer neutrons once the reactor is critical (i.e. In contrast, in a nuclear reactor, the rate at which the nuclear fuel undergoes fission is rigorously controlled using “control rods”: cylinders of neutron absorbing material that are inserted into the reactor core to absorb a portion of the neutrons released by the fission of the uranium-235 fuel. If the nuclear chain reaction is allowed to propagate unchecked, the rate of fission will increase rapidly as more and more neutrons are produced, resulting in a massive and nearly instantaneous release of energy and heat. Schematic representation of a nuclear chain reaction – energy release not shown.Īlong with a means of instigating nuclear fission in the reactor core, it is important to be able to control the rate at which the uranium atoms are undergoing fission. It is this chain reaction that provides the basis for nuclear research reactors and nuclear power, as it releases not only neutrons and fission products, but also large quantities of energy. This suggests the possibility that the neutrons released when one uranium-235 nucleus undergoes fission can induce the fission of up to three neighbouring uranium-235 nuclei, each of which will also release neutrons as it fissions, which can interact with other uranium-235 atoms, and so on, thus creating a self-sustaining nuclear chain reaction. Looking more closely at the figure above, it becomes evident that the nuclear fission of uranium-235 produces more neutrons (three in this example) than it consumes (one). Schematic representation of an induced nuclear fission event: uranium-235 captures a neutron and fragments into two smaller atomic nuclei, releasing neutrons and energy. Neutron bombardment can also be used to increase the rate of nuclear fission in nuclei that undergo this process spontaneously, such as uranium-235 (see Figure). 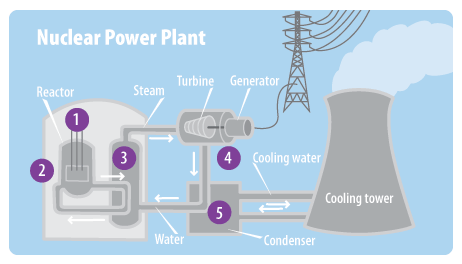
Nuclei that are susceptible to this “induced nuclear fission” include plutonium-239 and uranium-233. 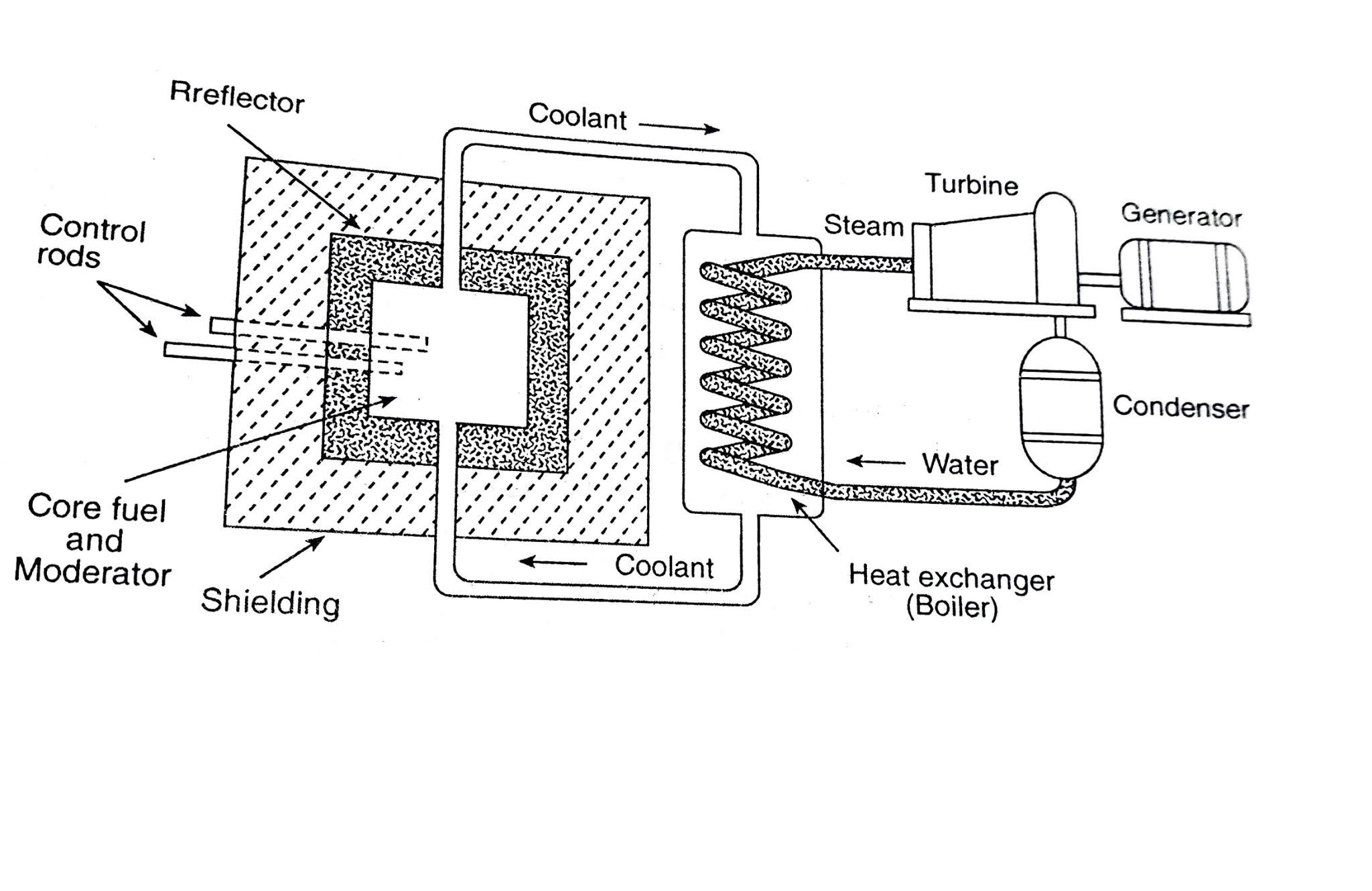
For example, a slow-moving or “thermal” neutron can be absorbed into the nucleus of an atom, rendering it unstable and causing it to fission instantly. In addition to nuclei that fission spontaneously, many other atomic nuclei can be induced to undergo nuclear fission by bombarding them with sub-atomic particles. 
The periodic table of the elements (from Heavy elements such as the actinoids are prone to nuclear fission. Despite being a spontaneous process, fission of these nuclei occurs very slowly in nature: the fission half-life of uranium-235 is 1.2 x 1017 years, which corresponds to one fission per gram per hour. Examples of materials in nature that spontaneously undergo nuclear fission are uranium-235 (an atomic nucleus containing 92 protons and 143 neutrons) and thorium-232 (90 protons and 142 neutrons). Nuclear fission only occurs in the heaviest elements of the periodic table: principally, the actinoid series, but more generally, those with atomic weights greater than 232 atomic mass units. This is often accompanied by the emission of neutrons, charged fragments such as alpha particles, and energy in the form of gamma or X-rays. Nuclear fission is a naturally occurring phenomenon during which an atomic nucleus is divided into two or more parts.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
